Maintaining blood vessel health – Solutions from nanoscience
Dr. May Azzawi, Vascular Physiology Group, School of Healthcare Science, Faculty of Science & Engineering, Manchester Metropolitan University.
The incidence of CardioVascular Disease (CVD) is on the rise worldwide, affecting more than 5 million people in the UK alone. The clinical manifestation of the disease, in the form of myocardial infarction and angina, is largely caused by poor blood supply due to narrowing of the arteries (such as due to the formation of blood clots and thickening of the arterial walls). Healthy blood vessels are thus important in ensuring that blood supply is matched to tissue demand.
Blood vessels are lined by an inner layer of endothelial cells, which release important mediators that maintain healthy vessel wall structure and function. An early sign of CVD is a ‘dysfunctional’ endothelial cell, where there is an imbalance in the release of vasodilator and vasoconstrictor mediators. This can cause narrowing of the vessel lumen and can ultimately lead to thickened arterial walls and accelerate the build-up of plaque (atherosclerosis). Changes associated with ageing, such as the accumulation of advanced glycation end-products and the generation of Reactive Oxygen Species (ROS), can affect mediator release from endothelial cells, contributing to cell dysfunction and leading to altered vessel function. It is also clear that impaired vessel dilator function can lead to reduced blood perfusion into tissues such as heart muscle, impairing the function of that tissue.
As part of an EU-funded project, in collaboration with Katholieke Universiteit, Leuven, Belgium, we at the school of Healthcare Science, Manchester Metropolitan University (MMU, Azzawi and Degens) are examining the role of antioxidants in the restoration of blood vessel function in ageing. The €6.5 million grant funding supports the joint Erasmus Mundus doctoral programme (MOVE-AGE- within MMU, KU Leuven and VU University, Amsterdam) which aims to maintain mobility in an ageing population, and prevent mobility loss in cases with age-related disease conditions, such as CVD.
The rapidly emerging field of nanotechnology offers attractive alternative options for the treatment and diagnosis of CVD. The unique properties, labelling and drug loading potential of some nanomaterials means that they can be used to target specific cells and tissues, offering new diagnosis and treatment strategies. One type of nanomaterial, cerium oxide, has unique antioxidant properties that are being explored for the treatment of conditions where there is increased ROS generation. The radical scavenging property of Ceria nanoparticles is due to the large number of surface oxygen vacancies, and has been described to be more efficient than the biological antioxidant, superoxide dismutase.
In collaboration with MMU chemist, Dr. Debra Whitehead, we are exploring the use of cerium oxide nanoparticles, as a novel therapeutic strategy for the treatment of vascular disease [Figure 1].1 Early work by our interdisciplinary group has demonstrated that the rapid uptake of nanoparticles, by endothelial cells that surround the blood vessels [Figure 2], may be detrimental to their function (EPSRC bridging-the-gap funding).2,3 The large surface area to volume ratio of the nanoparticles (due to their small size) means that they may be highly reactive, interacting with cellular organelles and interfering with mediator release from endothelial cells. Furthermore, nanoparticles of certain material composition, such as quantum dots, may interfere with nitric oxide donor drugs, such as sodium nitroprusside, used in the treatment of CVD.4
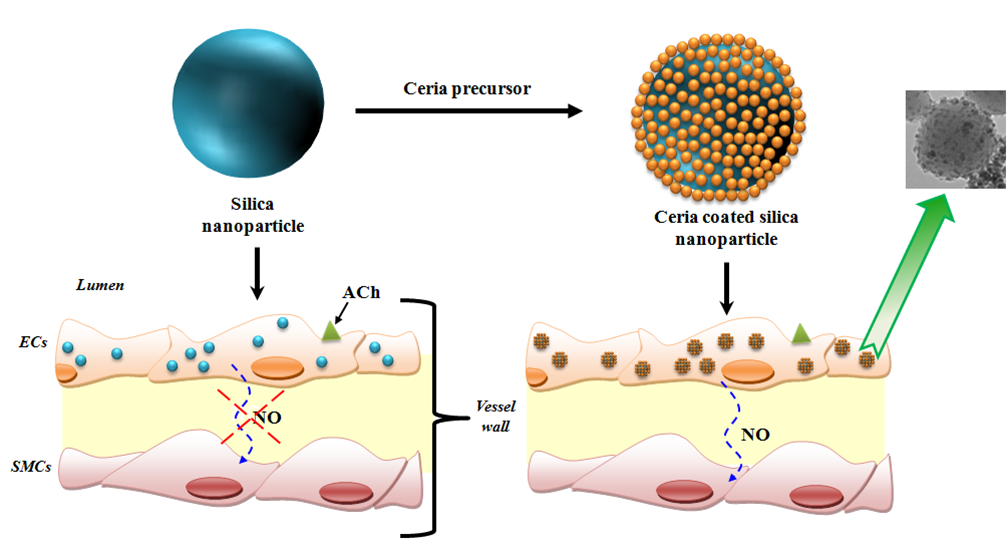
Figure 1
Graphical representation of the influence of nanoparticles on dilator responses of vessels. Nanoparticle uptake by endothelial cells lining the blood vessel. While silica nanoparticles reduce endothelial dependent (Acetylcholine) dilator responses, their surface modification with ceria nanoparticles leads to improvement in dilator responses (ACh, Acetylcholine; ECs, endothelial cells; NO, Nitric oxide; SMCs, smooth muscle cells). Dr. Asima Farooq, Postdoc. 1
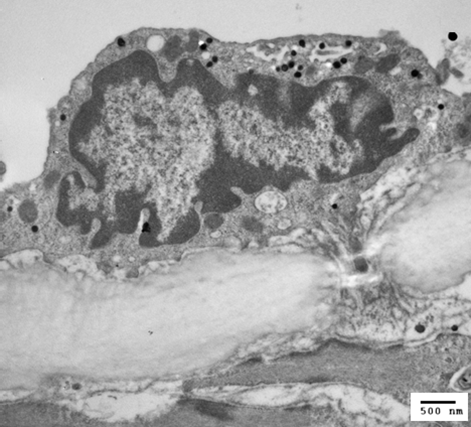
Figure 2: Transmission electron micrograph showing silica nanoparticle uptake (100 nm) by an endothelial cell lining a mesenteric artery after a 30-minute incubation period. The nanoparticles are identified as dark spherical objects within the cytoplasm of the cell (magnification x25,000).Preparation of EM sample, Dr. Carolyn Jones (Maternal and Foetal Health Research Group, University of Manchester).
Nanoparticles can penetrate blood vessels via two modes of exposure: Intravascular and Extravascular. Intravascular exposure takes place mainly through intravenous injection of ‘engineered’ nanoparticles, used for medical diagnostic and therapeutic purposes [Figure 3]. Extravascular exposure, on the other hand, takes place when nanoparticles are inhaled, ingested, or absorbed through the skin (examples include nanoparticles found in air pollutants and chemical reagents). As these nanoparticles are absorbed through the mucosa, they can translocate through extracellular tissues and interstitial spaces, into the outer adventitial layer of blood vessels, passing through the entire vessel wall and reaching into the endothelial cell layer.5
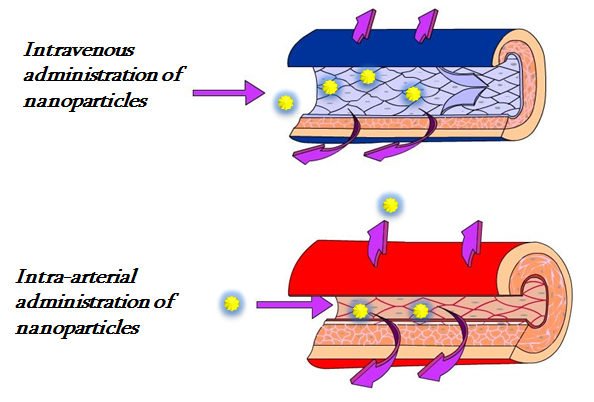
Figure 3: Illustration of blood vessel exposure to nanoparticles. Intravascular exposure to nanoparticles (yellow spheres) is one mode of exposure (intravenous or intra-arterial). Illustration by Mo AlKattan.
Using a state-of-the-art ex vivo model of vascular function, our group is testing a range of nanoparticles, of various material composition and surface coatings, in collaboration with Alexander Seifalian, Professor of Nanotechnology and Regenerative Medicine, at University College London. The aim of our group is to inform the use of safer, more biocompatible nanoparticles, and the use of appropriate nanoparticle coatings for the protection and restoration of blood vessel function. We will work with translational and cardiovascular scientist, Professor Yvonne Alexander and senior research fellow, Dr Fiona Wilkinson (Cellular biologist, MMU) towards testing novel diagnostic tools and treatments for CVD.
With the rapid growth of engineered nanomaterials, for the diagnosis and treatment of CVD, there is now an ever-increasing need to screen these nanoparticles and ensure their safety and biocompatibility. Moreover, there is a growing need to identify non-animal technologies that help replace and minimise the requirement for animal tissues in toxicity testing. MMU is now at the forefront of exploring non-animal technologies, and in collaboration with industrial partners, we aim to develop a novel 3D bioengineered cardiovascular model for toxicity testing (Azzawi & Hidalgo [Bioengineer]). Such models will allow the screening of novel therapeutic drugs, including nanomedicines, and will reduce the need for animal experimentation thus supporting the government agenda of the 3Rs (Replacement, Reduction and Refinement) for animal welfare.

Photo
Dr. May Azzawi, is a senior lecturer in the School of Healthcare Science, Healthcare Science Research Institute, Manchester Metropolitan University, and an Executive member of the British Society for nanomedicine.
REFERENCES
- Farooq A, Mohamed T, Whitehead W, Azzawi M. Restored vasodilator responses after infusion of ceria coated silica nanoparticles, ex vivo. J Nanomed Nanotechnol, 2014; 5:195-199.
- Akbar N, Mohamed T, Whitehead D, Azzawi M. Biocompatibility of amorphous silica nanoparticles: Size and charge effect on vascular function, in vitro. Biotechnol Appl Biochem. 2011; 58(5):353-62.
- Farooq A, Whitehead D, Azzawi M. Attenuation of endothelial dependent vasodilator responses, induced by dye encapsulated silica NPs, in-vitro. Nanomedicine, 2014; 9:413-25.
- Shukur A, Rizvi S, Whitehead D, Seifalian A and Azzawi M. Altered sensitivity to nitric oxide donors, induced by intravascular infusion of quantum dots, in murine mesenteric arteries. Nanomedicine-NBM 2013; 9(4): 532-539.
- Azzawi M. ‘The nanotoxicological influence of nanoparticles; with special reference to the vasculature’. In: Current advances in the medical application of nanotechnology. Ed. M. Slevin. Bentham Publications, 2012.